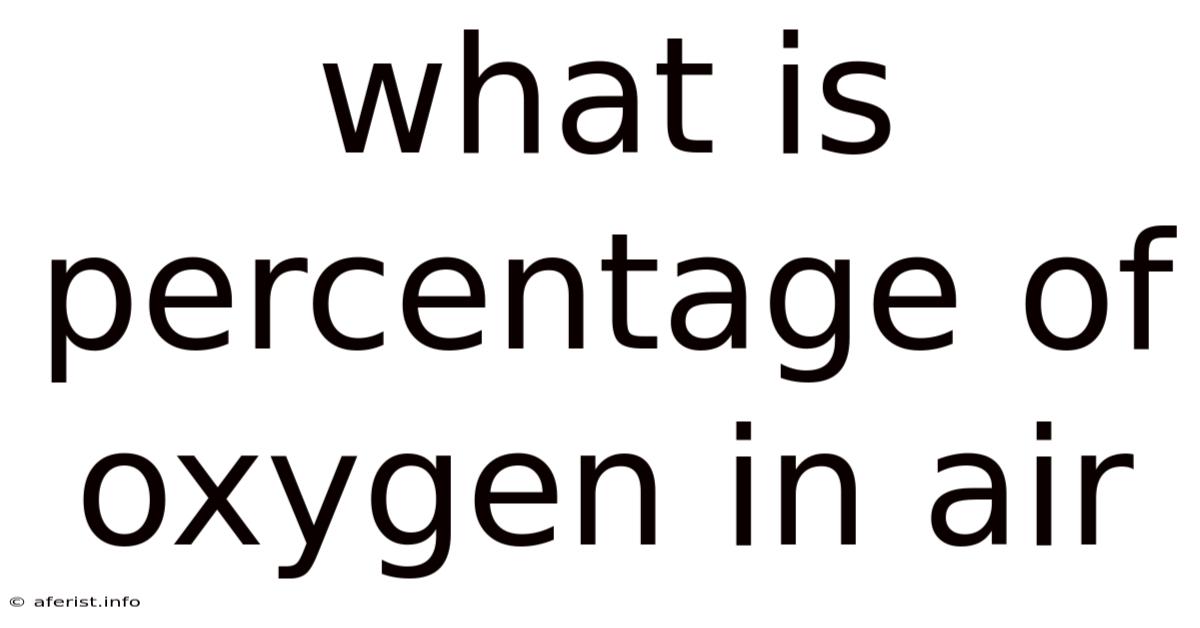What is the Percentage of Oxygen in Air? A Deep Dive into Atmospheric Composition
The air we breathe is a complex mixture of gases, and understanding its composition is crucial to comprehending various aspects of our environment, from human health to climate change. But while a simple answer is readily available, a deeper exploration reveals fascinating insights into the dynamics of our atmosphere and its significance for life on Earth. On top of that, a fundamental question that arises is: what is the percentage of oxygen in air? This article will walk through the precise percentage of oxygen in the air, explore the factors influencing its concentration, discuss the importance of this vital gas, and address some common misconceptions That's the part that actually makes a difference. But it adds up..
Introduction: The Vital Role of Oxygen
Oxygen (O₂) is undeniably the most crucial component of air for most living organisms. Worth adding: it's the essential element in the process of cellular respiration, where energy is extracted from food molecules. Without a sufficient supply of oxygen, life as we know it would cease to exist. On the flip side, the percentage of oxygen in the air, therefore, is a key indicator of the health and habitability of our planet's atmosphere. This percentage isn't static; it varies slightly depending on location, altitude, and time of day, but a general average provides a valuable benchmark The details matter here..
The Percentage of Oxygen in Air: The Standard Value
The standard percentage of oxygen in dry air at sea level is approximately 20.In practice, 95% or 21%. This figure is often rounded to 21% for simplification, but the more precise measurement is crucial for scientific accuracy. don't forget to remember that this is the percentage by volume; other methods of measuring gas concentration, such as by weight or mole fraction, would yield slightly different numerical results.
The remaining 78% of dry air is primarily nitrogen (N₂), with trace amounts of other gases like argon (Ar), carbon dioxide (CO₂), neon (Ne), helium (He), and methane (CH₄). These trace gases, while present in smaller quantities, play significant roles in various atmospheric processes, including climate regulation and the ozone layer. Water vapor is also a significant component of air, but its concentration is highly variable and can range from near zero to several percent, depending on temperature and humidity Most people skip this — try not to. And it works..
Factors Influencing Oxygen Concentration: A Dynamic Atmosphere
While the average percentage of oxygen in air remains relatively stable, several factors can cause localized variations:
-
Altitude: The concentration of oxygen decreases with increasing altitude. This is because gravity pulls heavier gases, like oxygen and nitrogen, closer to the Earth's surface. At higher altitudes, the proportion of oxygen in the air is noticeably lower. Mountaineers, for example, face the challenge of reduced oxygen availability at high elevations, necessitating supplemental oxygen supplies for extreme climbs Most people skip this — try not to. Took long enough..
-
Pollution: Human activities, particularly the combustion of fossil fuels, can indirectly affect oxygen levels. While the process doesn't directly consume oxygen, the release of pollutants such as carbon monoxide (CO) and particulate matter can reduce the air's quality and impair oxygen uptake in the lungs. Adding to this, deforestation contributes to reduced oxygen production as fewer trees are available to perform photosynthesis.
-
Photosynthesis and Respiration: The continuous interplay between photosynthesis (oxygen production by plants) and respiration (oxygen consumption by animals and plants) matters a lot in maintaining the balance of oxygen in the atmosphere. These two processes are essential to the carbon-oxygen cycle, ensuring a relatively stable oxygen concentration over the long term. Seasonal variations in plant activity can cause slight fluctuations in atmospheric oxygen levels The details matter here..
-
Oceanic Processes: The oceans act as a massive reservoir of dissolved oxygen. The exchange of oxygen between the atmosphere and the ocean plays a significant role in maintaining the global oxygen balance. Ocean currents and changes in water temperature affect the rate of this exchange, indirectly influencing the atmospheric concentration of oxygen Worth keeping that in mind..
-
Geographic Location: Local variations in plant life, industrial activity, and weather patterns can cause small differences in oxygen levels across various geographic locations. Densely forested areas typically exhibit slightly higher oxygen levels compared to urban areas with significant pollution Worth keeping that in mind..
The Importance of Oxygen: Sustaining Life on Earth
The significance of oxygen in maintaining life on Earth cannot be overstated. Its role extends far beyond simply facilitating cellular respiration:
-
Cellular Respiration: Oxygen is the final electron acceptor in the electron transport chain, a crucial part of cellular respiration. This process generates the energy (ATP) needed for virtually all cellular activities, including growth, repair, and movement.
-
Oxidation: Oxygen plays a vital role in many biochemical reactions, including oxidation processes that break down food molecules and detoxify harmful substances.
-
Ozone Layer Formation: Oxygen in the upper atmosphere forms ozone (O₃), which absorbs harmful ultraviolet (UV) radiation from the sun, protecting life on Earth from its damaging effects That alone is useful..
-
Combustion: Oxygen is essential for combustion, which is used extensively for energy production in various applications That's the part that actually makes a difference. Practical, not theoretical..
Understanding the Numbers: Percentages, Partial Pressures, and Concentrations
The percentage of oxygen (20.This value is crucial in understanding oxygen transport in the body, as it determines the rate of oxygen diffusion across cell membranes. The partial pressure of oxygen is the pressure exerted by oxygen molecules in a mixture of gases. Because of that, 95 liters would be oxygen. Even so, it's equally important to understand the concept of partial pressure. 95%) represents the volume fraction of oxygen in dry air. So in practice, in a sample of 100 liters of dry air, approximately 20.At sea level, the partial pressure of oxygen is approximately 160 mmHg (millimeters of mercury). Understanding both the percentage and the partial pressure provides a more comprehensive view of oxygen availability Most people skip this — try not to..
Frequently Asked Questions (FAQ)
-
Q: Does the percentage of oxygen in air change over time?
A: While the long-term average remains relatively stable, small fluctuations occur due to the factors mentioned above (photosynthesis, respiration, pollution, etc.). Significant changes in atmospheric oxygen levels would have catastrophic consequences for life on Earth.
-
Q: Is it possible to have too much oxygen in the air?
A: Yes, although rare in natural environments, significantly elevated oxygen levels can be toxic. Increased oxygen concentration can lead to oxidative stress, damaging cellular components and contributing to various health problems.
-
Q: How is the percentage of oxygen in air measured?
A: Various methods are used, including gas chromatography, which separates and quantifies the components of a gas mixture. Other techniques involve electrochemical sensors and spectroscopic methods.
-
Q: Why is the percentage of oxygen in air considered crucial for human health?
A: Because oxygen is essential for cellular respiration, providing the energy required for all bodily functions. Insufficient oxygen levels can lead to hypoxia (oxygen deficiency), causing various health problems, ranging from mild fatigue to severe organ damage and death.
Conclusion: The Ever-Present Importance of Oxygen
The percentage of oxygen in air, approximately 20.95%, is a fundamental constant that underpins life on Earth. Practically speaking, while this value remains relatively stable, fluctuations can occur due to a complex interplay of natural and anthropogenic factors. Understanding these variations and the role of oxygen in various atmospheric processes is vital for addressing environmental challenges and safeguarding the health of our planet and its inhabitants. Continued research and monitoring of oxygen levels are essential to ensuring a healthy and sustainable future. Appreciating the delicate balance of this vital gas in our atmosphere underscores the need for responsible stewardship of our environment. Further investigation into the involved dynamics of the oxygen cycle will continue to refine our understanding of this crucial aspect of our planet's life support system Worth keeping that in mind. Surprisingly effective..